- Log in to post comments
In eukaryotic cells, histone protein octamers serve as the structural scaffold for genomic DNA to wrap around to form nucleosomes – the basic units of chromatin. The canonical octamer consists of two copies of H2A, H2B, H3 and H4, while diverse histone variants could substitute the canonical ones to serve special functions.
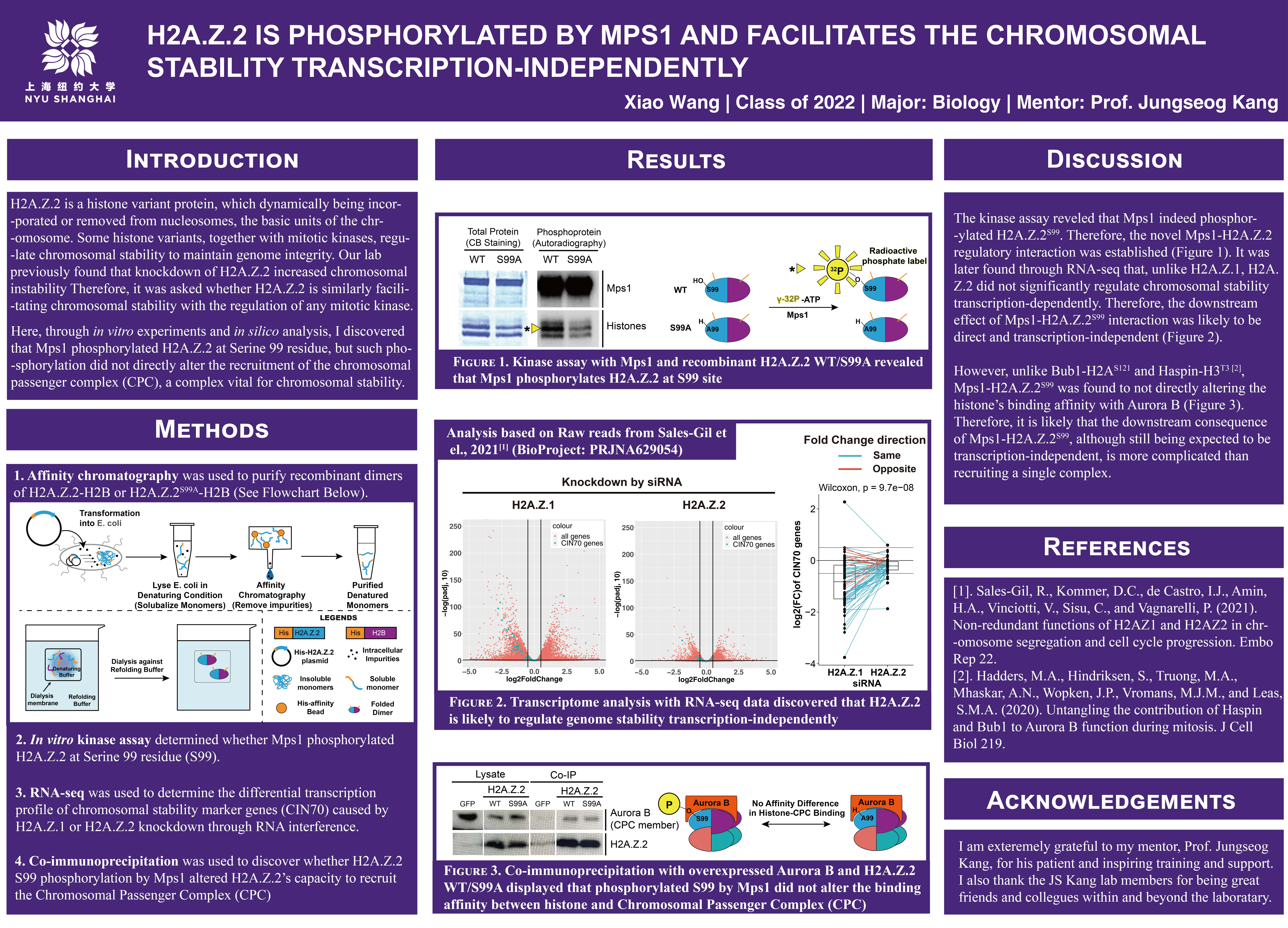
Histone variant H2A.Z.2 has been reported to directly facilitate chromosome segregation in a transcriptional-independent manner. Somatic cells divide through mitosis, and H2A.Z.2’s direct involvement in chromosome segregation during mitosis is vital for genome integrity. Otherwise, aneuploidy will occur and in the worst case, may lead to cancer. Therefore, it is with great biological significance to understand the function and regulation of H2A.Z.2 in chromosome segregation.
Multiple mitotic kinases regulate the progression of mitosis through phosphorylating their substrates. Therefore, we asked whether H2A.Z.2 would be regulated by phosphorylation as well. H2A.Z.2 has been reported to be phosphorylated at the serine 99 (S99) site, but the kinase that contributed to this phosphorylation remained unknown. Our lab has previously discovered that H2A.Z.2 interacted with mitotic kinase Monopolar spindle 1 (Mps1). And my project attempted to verify Mps1’s phosphorylation on H2A.Z.2 at S99 site and investigate the biological significance of such phosphorylation during mitosis.
To verify the phosphorylation, an in vitro kinase assay was performed. I prepared soluble recombinant H2A.Z.2-H2B wildtype and H2A.Z.2(S99A)-H2B mutant dimers by separately expressing the monomers in E. coli. The S99 site in the mutant dimer was changed into alanine so that its phosphorylation substrate activity was abolished. Monomers were purified through His-tag affinity chromatography in denatured conditions and mixed to form biologically-active dimers by dialyzing in renaturing buffer. The kinase assay results confirmed a weaker phosphorylation signal on H2A.Z.2(S99A)-H2B mutant comparing to the wildtype, and it served as strong evidence proving Mps1’s phosphorylation of H2A.Z.2 at S99 site. To further confirm it, the phospho-S99-specific antibody is under production and will be used to visualize phosphorylation signal in a second kinase assay.
To investigate its biological significance, we questioned whether phospho-S99 directly alters the interaction between H2A.Z.2 and the CPC complex, as in the case of H2A.Z.2 acetylation. Therefore, I performed a co-immunoprecipitation assay between HA-Aurora B and Myc-H2A.Z.2/Myc-H2A.Z.2(S99A) that were overexpressed in HeLa cell culture to evaluate the direct or indirect binding between the histone and the CPC (Aurora B is a key component in the CPC). However, no significant difference in binding affinity was observed between H2A.Z.2/H2A.Z.2(S99A) and the Aurora B. Likely, Mps1’s phosphorylation on H2A.Z.2 did not directly regulate mitosis through facilitating the recruitment of the CPC complex.
To summarize, I confirmed that H2A.Z.2 is very likely to be phosphorylated by the mitotic kinase Mps1 at the S99 site, while such phosphorylation could not regulate mitosis directly by recruiting the CPC. It raises further investigation on the function of S99 phosphorylation in the future to study mitosis.
*Further experiments to investigate the impact of kinase inhibitors on H2A.Z.2 overexpression have started by paused due to the pandemic. Results will be included if finished.
Comments
- Log in to post comments


It is a good and interesting study. Two comments. First, it is better to include an illustration that explains how centromeric histone variants play a role in chromosome segregation with other players like Mps1 and CPC. People like to see a big picture for understanding. Second, maybe you explain the difference of two isoforms, H2A.Z.1 and H2A.Z.2?